Popular Links
Latest News See All News
Events See All Events
Featured Products
FCI is proud to present its latest innovations!
FCI Ophthalmics
State-of-the-Art Ophthalmic Surgical Devices
Since its inception in 1984, FCI has become a world leader in the development of ophthalmic surgical devices. The products manufactured by FCI include a full range of lacrimal stents and tubes, eyelid implants and weights, orbital implants, cataract devices, vitreoretinal devices, and dry eye products.
DRY EYE
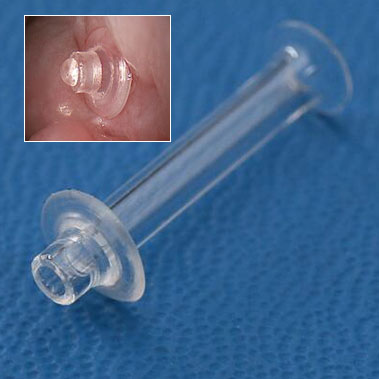
Perfectly sized punctum plugs for your patients’ dry eyes.
FCI Ophthalmics has been a leader in the research, design and manufacturing of highly effective tools for the treatment of dry eyes including multiple types of punctal plugs. Snugplug® a result of extensive research in this area, is a unique tool that eliminates sizing, simplifies stocking, avoids dilation and facilitates insertion in one step.

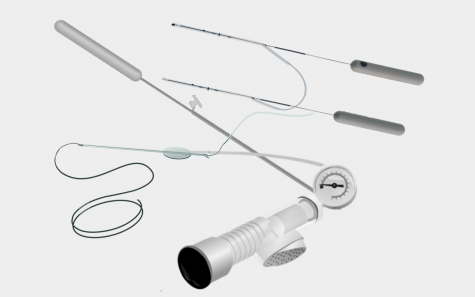
LACRIMAL
A wide range of flexibility in your surgical approach
The comprehensive treatment of nasolacrimal duct obstruction and stenosis is FCI Ophthalmics’ core business. As a world leader in the R&D of lacrimal devices we provide bicanalicular and monocanalicular stents and tubes.
EYELID REPAIR
An array of eyelid devices
FCI Ophthalmics is dedicated to helping physicians provide both surgical and non-surgical solutions, including eyelid implants and external eyelid weights, for the treatment of eyelid difficulties such as lagophthalmos and ptosis.

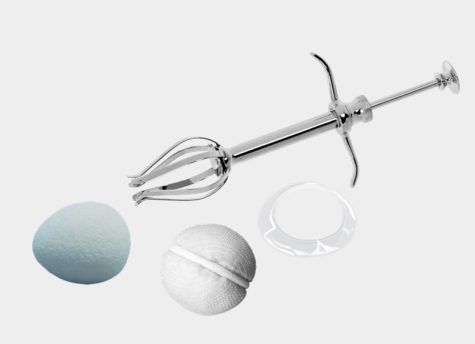
ORBITAL
Improved biocompatility of our implants
FCI Ophthalmics orbital implants provide highly effective treatment options to perform enucleation and evisceration procedures. Among other ophthalmic surgical devices, FCI offers silicone implants, bioceramic implants and OSMED Tissue Expanders.
CATARACT
Your complex cases converted into simple cases
As a world leader in the development of cataract surgery devices, FCI Ophthalmics has worked with reputable manufacturers. From CTRs to the Mackool capsular bag hooks, you can trust FCI to be your complete supplier.
VITREORETINAL
Our Focus is on your success
For decades FCI has developed a wide range of retina products such as: laser probes, vitreoretinal instruments, vitrectomy lenses, backflush and cannulas. FCI expertise led to the development of many innovative and exclusive devices.
“Historically, repair of either canaliculus involved bicanalicular intubation with intranasal retrieval of stent tubing, often under general anesthesia. There are now a number of commercially available stent products that allow a wide range of flexibility in surgical approach.”
Tamara R. Fountain, MD – Chapter 5 “Management of Canalicular Trauma”
Manual of Oculoplastic Surgery by Mark R. Levine (2010)